Innovative medical device which aims to revolutionise the detection of lazy eye in children
Business overview
| Location | London, United Kingdom |
|---|---|
| Social media | |
| Website | www.irissmedical.com |
| Company number | IRISSMedical |
| Incorporation date | 7 Jun 2011 |
Idea
Introduction
An innovative, patent protected medical device that performs automatic eye tests aimed at the early detection and prevention of lazy eye in children. Lazy eye is the impairment of vision in one eye and is the leading cause of preventable blindness in people under 40.
Even in the 21st century, eye specialists and doctors rely on manual techniques to conduct examinations for the diagnosis of conditions such as squint, for fitting contact lenses and for assessing eyes prior to lid surgery. Some of the techniques still in use today were developed over a century ago. They require considerable skill and experience in order to achieve the best outcome. The result is that 3% of children suffer from lazy eye which had been missed, and 16% of contact lens wearers drop out due to poor fitting.
IRISS Medical has developed a device that automatically provides accurate eye measurements. It is the digital version of those techniques and aims to address these problems. The device was launched in late 2014

Intended impact
Lazy eye (amblyopia) affects approximately 3% of the child population. Its chief cause, squint, is a complex condition which is missed in many children and, as a result, they may grow up to develop a permanent vision impairment in one eye. Early identification and treatment of squint is essential for successful treatment, but a significant proportion of squint cases in children are undetected and go untreated.
Contact lenses is an area in which most of the available products are aimed at patients whose eye parameters fall within a certain "norm" both in terms of the physical fit of the lens and the optics that are required. The result is that many patients suffer from poor fitting or simply drop out from wearing contact lenses.
The IRISS device hopes to revolutionise the way in which conditions such as squint are detected; the way contact lenses are fitted and the way other measurements of the eyes are carried out.
Instead of using millimeter rulers and other manual techniques, the IRISS device automatically provides accurate measurements of the eyes. This will enable doctors, optometrists and health screeners to:
- Identify and treat a much larger proportion of the 3% of children
worldwide who develop squint and lazy eye;
- Better fit contact lenses for the 27% of the population whose
eye parameters fall outside the norm and to drastically reduce the
16% of contact lens wearers who drop out.
- Significantly enhance the accuracy in measuring key eye features such as pupils and lids as part of routine or surgical work;
The IRISS device has the potential to become the new Gold Standard for external eye measurements and to be widely used by clinicians throughout the world.
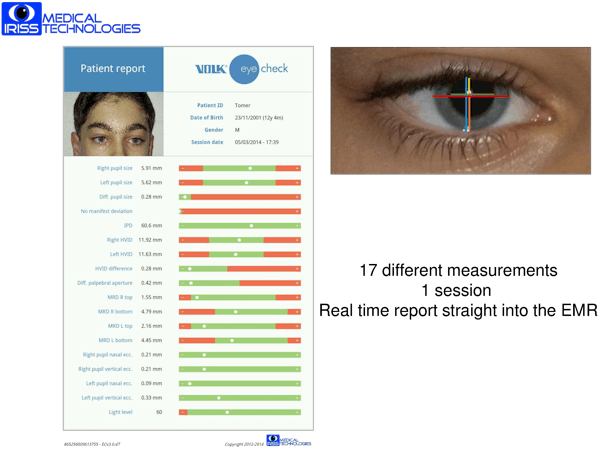
Substantial accomplishments to date
IRISS is no longer a pre-revenue company, and has recently launched its product on the market. Founded in June 2011, to date IRISS Medical has -
- Developed, commercialised and launched the device, branded as
Volk Eye Check.
- Achieved early revenue by beginning to sell devices as of late 2014.
- Partnered with US based Volk Optical Inc., global leader in
ophthalmic diagnostic lenses, for the marketing and distribution of
the device.
- Been granted two international patents with a third patent pending.
- Received regulatory clearance from the US FDA and the CE Mark
for the European Union, clearing the way to selling in the US and in
Europe.
- Been awarded ISO 13485 accreditation - the international quality standard for medical device companies.
- Published 3 scientific posters at international medical conferences.

Monetisation strategy
IRISS aims to sell the Volk Eye Check device to optometrists and eye doctors as well as to GPs and screening programmes.
The business model consists of two parts:
1. Selling the device into the market, via its partnership with Volk Optical. This involves distribution channels into the optometric market. This part of the business model has already begun, with IRISS achieving early revenue as of late 2014.
2. For contact lens specialists, IRISS aims to receive revenue on a pay- per-use basis. This is because the device can provide on-going recommendations as to the best fit lens. Therefore IRISS will look to provide both a device and a service to contact lens specialists.
Use of proceeds
Following the commercialisation of the Volk Eye Check device, IRISS now needs to support the launch of the device and accelerate its acceptance in the marketplace.
Proceeds will be used to scale up and support the sales and marketing efforts by:
- Creating the necessary sales resources to support its partners across the world;
- Conducting trials and studies for further validation and market recognition;
- Establishing presence in key trade shows in the US and Europe.
Market
Target market
Vision develops during early childhood and is usually complete by 6 to 8 years of age. Lazy eye is a condition whereby the vision in one eye is reduced, because vision has not developed normally, and this cannot be corrected with glasses. Lazy eye has a prevalence of approximately 3% and is the leading cause of preventable blindness in people under the age of 40 - more than all other causes combined. Several underlying conditions lead to lazy eye, of which a major one is "squint" (strabismus). Squint is the misalignment of the two eyes which has a prevalence of approximately 4% in young children across the world. Squint and lazy eye are treatable in most cases if detected early enough and provided the appropriate treatment is given. If lazy eye is not treated by the age of 7 or 8, it becomes irreversible and the damage to vision in that eye is permanent.
There are 125 million contact lens wearers worldwide. Drop-out rates among contact lens wearers vary between 16%-30%, depending on demographics. A key reason for that is that "standard" contact lenses are designed to fit approximately 75%-80% of the population, with the rest requiring "non-standard" fitting.
The target market for the IRISS device consists of the clinicians who are responsible for detecting and treating squint; for performing basic eye examinations; and clinicians who fit contact lenses. These include:
- Eye care specialists, such as ophthalmologists (eye doctors) and optometrists (perform basic eye exams and fit glasses/ contact lenses);
- GPs, family practice doctors and paediatricians;
- School and community screening programmes as well as health visitors and child well-being clinics.
There are approx. 450,000 optometrists and ophthalmologists worldwide as well as approx. 2 million GPs and family practice doctors who in many cases are the primary care contact for eye health. Therefore, the total addressable market for the IRISS device consists of approx. 2.5 million clinicians worldwide.
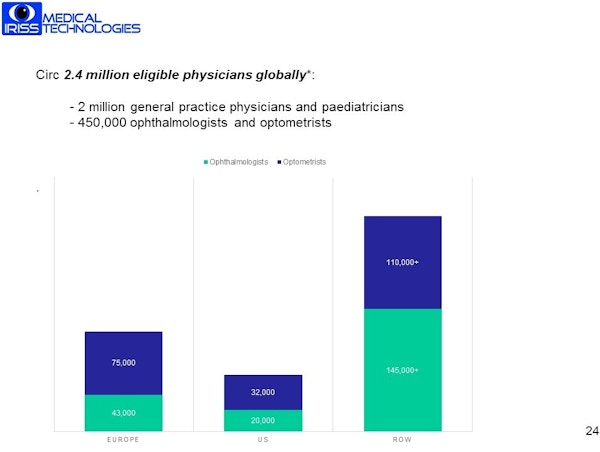
Characteristics of target market
There are 650 million children worldwide under 5 served by the 2 million GPs and family doctors as well as the 450,000 eye specialists. Additionally, 125 million wearers of contact lenses are served by these same specialists.
Currently, eye specialists rely on manual techniques to perform examinations to detect squint and other conditions, such as those related to pupils and lids. We believe that these specialists would greatly benefit from an objective, accurate and fast device that provides them with measurements and data regarding these conditions. It should assist them in making better clinical decisions and, in some cases, save them time.
We believe that contact lens specialists will greatly benefit from reducing the drop-out rates of contact lens wearers and from saving patient "chair time" in what is often a protracted fitting process.
GPs and family practice doctors are not skilled at correctly identifying squint and this will enable them to greatly reduce the number of missed squints and lazy eye.

Marketing strategy
Early on in its development, IRISS decided to partner with a major eye care brand to bring the device to the market. US based Volk Optical, the market leader in ophthalmic diagnostic lenses, was identified and is IRISS's partner for the sales and marketing of the device.
The advantage to IRISS is that Volk is a strong brand, widely recognised by eye care specialists throughout the world, and has distribution channels in most countries around the globe.
IRISS is already executing on its plans to reach the market via its partner, initially in the UK, US and Europe and later on in Asia, Latin America, Africa and Australia.
Once the device achieves penetration and acceptance with eye specialists, IRISS will look to expand its distribution to the other market segments - GPs and family practice doctors.
Competition strategy
As far as we are aware, the IRISS device, Volk Eye Check, is unique and novel. We don't believe that any other device provides the measurements and data that it provides. Volk Eye Check is protected by two patents, with a third patent pending. The technology underlying the Volk Eye Check device consists of sophisticated mathematical algorithms, which are the patented part, combined with proprietary IP for in-camera integration. We believe that the barriers to entry for producing such a device are therefore high.
In addition, IRISS's objective is to penetrate the market and achieve recognition quickly, so that if and when a similar/ competitive device reaches the market, it will encounter a well-entrenched device. Brand recognition of the Volk Eye Check would also be a competitive advantage against any newcomer.
Open an account to get access to the team members of IRISS Medical
Already have an account? Log in
To comply with financial regulations, we can only show full campaign details to registered users.
Open an account to get access to the IRISS Medical campaign updates
Already have an account? Log in
To comply with financial regulations, we can only show full campaign details to registered users.
Open an account to get access to the list of investors in the IRISS Medical campaign
Already have an account? Log in
To comply with financial regulations, we can only show full campaign details to registered users.
Open an account and verify your identity to get access to the IRISS Medical discussion
Already have an account? Log in
To comply with financial regulations, we can only show full campaign details to registered users.
Open an account and verify your identity to get access to the IRISS Medical pitch deck and other documents
Already have an account? Log in
To comply with financial regulations, we can only show full campaign details to registered users.
